 1b) recently, yet so far has scarcely been discussed. 3 An alternative to this classical approach that involves a positively charged halogen, X +, formally a halonium ion or halogen( I), as a halogen bond donor has also been introduced ( Fig. 3 The past two decades of halogen bonding research has consequently focused on the investigation of strong and thus comparably easy to detect complexes, in which a large electrophilic area on the halogen, typically called a σ-hole, has been promoted by perfluorination of the nearby carbon atoms ( Fig. 2 Being a weak interaction, halogen bonding is typically difficult to detect, especially in disordered phases. 1 It resembles hydrogen bonding and has accordingly been proposed to be applicable as a complementary tool for the rational modulation of molecular recognition events in chemistry and biology. Introduction Halogen bonding (XB) is the directional, non-covalent interaction of an electron-poor region of a halogen and a Lewis base. He moved to Uppsala University in 2017, and carries out research on halogen bonding, NMR method development, natural product chemistry and medicinal chemistry. Carlomagno, in 2009 he initiated his independent career at the University of Gothenburg, Sweden, where he was appointed professor of organic chemistry in 2017. Following an additional postdoctoral project in structural biology under the mentorship of Prof.  
Perrin (University of California, USA), he moved to the Max Planck Institute for Biophysical Chemistry to learn NMR spectroscopy from Prof. After postdoctoral research in physical organic chemistry with Prof. Gogoll (Uppsala University, Sweden, 2004). Mate Erdelyi graduated at Semmelweis University, Hungary, and obtained his PhD in organic chemistry under the guidance of Prof. Her current research interests are focused on three-center halogen bonds and their applications in synthetic organic chemistry. Mate Erdelyi at Uppsala University, Sweden. In 2018, she joined the research group of Prof. Lotta Turunen received her PhD in 2017 from the University of Jyväskylä, Finland, under the supervision of Prof. Following the discussion of the nature and properties of halonium ions’ halogen bonds, this tutorial review provides an overview of their current applications to stimulate future developments. The three-center halogen bond modulates the reactivity of halonium ions and is hence a useful tool for synthetic organic chemistry. The three-center halogen bond of halonium ions shows distinct differences in its properties from coordinative bonds of transition metals and is therefore applicable as a complementary synthon in supramolecular chemistry. 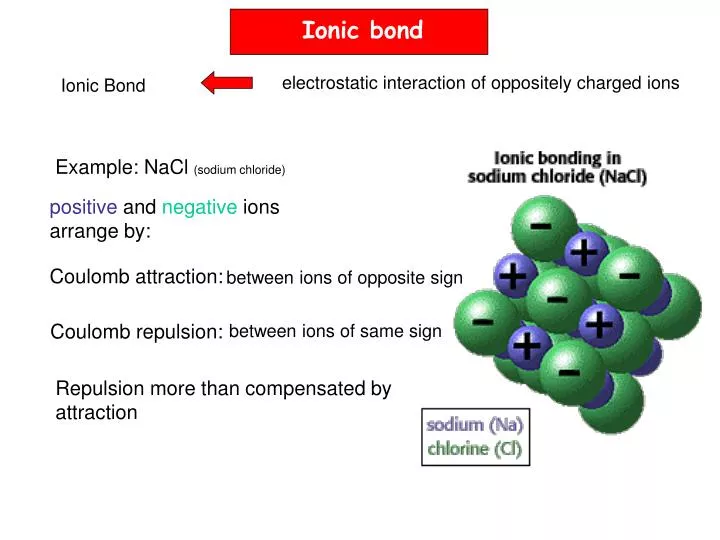
In analogy to the + hydrogen bonds, which are at times entitled as short and strong bonds, the + halogen bonds of halonium ions show Bondi normalized interatomic distances of 0.6–0.7 and possess both charge transfer and electrostatic characteristics. 
The interaction presumes the formation of three molecular orbitals and is accordingly typically entitled as a three-center halogen bond. By accepting electrons in both lobes of their empty p-orbital, they are capable of simultaneously interacting with two Lewis bases. Due to their electron deficiency, halonium ions act as particularly strong halogen bond donors. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
